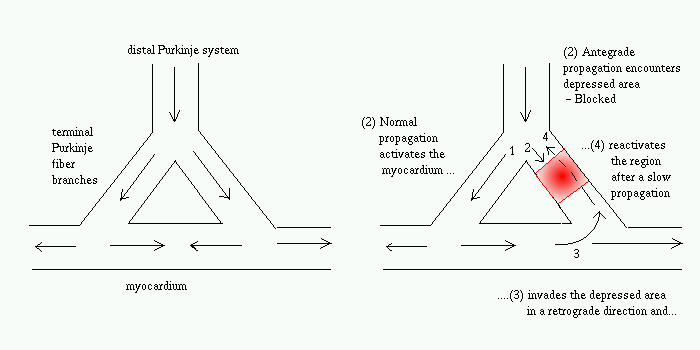
FIG. Propagation of normal action potential (left) and conditions for reentrant excitation (right)
My Collaborators: David
J. Christini and Kenneth M. Stein (Weill Medical College of Cornell
University, New York)
Pacing (rapid low-voltage stimulation from an implanted device) is regularly used to terminate cardiac arrhythmias such as Ventricular Tachycardia. Unfortunately, the mechanism for termination is not clearly understood. In this study we looked at the specific case of anatomical reentry (re-entry around non-conducting ventricular scar tissue) to look at the spatio-temporal sequence of steps when a pacing treatment is either succesful or unsuccessful.
A Model for Reentry (Schmitt
& Erlanger, 1928)

FIG. Propagation of normal action potential
(left) and conditions for reentrant excitation (right)
How to terminate reentry by pacing
stimuli ?
Each pacing wave splits into two branches while
traveling around the reentry circuit - anterograde (along the direction
of the rentrant wave) and retrograde (opposite to the direction of the
reentrant wave).
Pacing can result in :
Pacing-induced elimination of
Ventricular Tachycardia (VT)
Several factors influence the ability of extrastimuli
and/or rapid pacing to interact with VT:
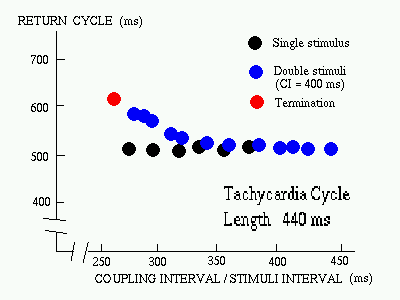
FIG. The response pattern of a VT to the delivery of single and double extrastimuli (Josephson [1993]) |
Why multiple stimuli
?
As there are a number of conditions to be satisfied before reentry is successfully terminated - a single extrastimuli is rarely sufficient. Double stimuli is often used - where the first extrastimulus is used only to "peel back" refractoriness to allow the second extrastimulus to interact with the circuit more prematurely. |
Figure-of-Eight Reentry
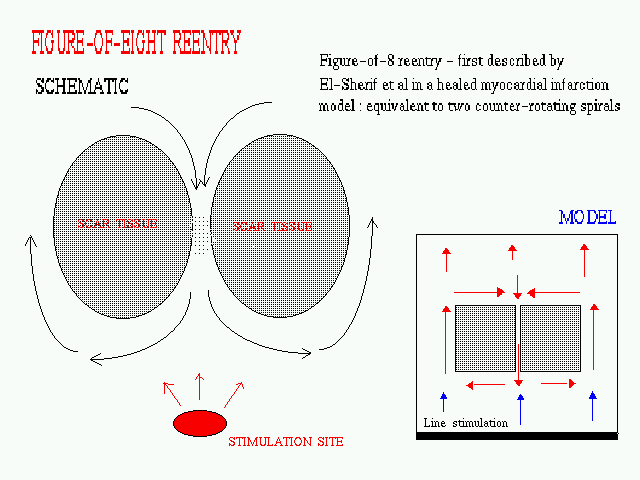
FIG. A schematic diagram of a anatomical
figure-of-eight reentry being paced from a stimulation site at the apex
of the ventricles. The inset shows the design of the model used for our
2-D simulatioms, the square patches representing non-conducting scar tissue.
The stimulation appears as a plane wave coming from the bottom of the region
(this represents a wavefront propagating outward from the ventricular apex).
Eliminating reentry in 2-D is
very different from 1-D
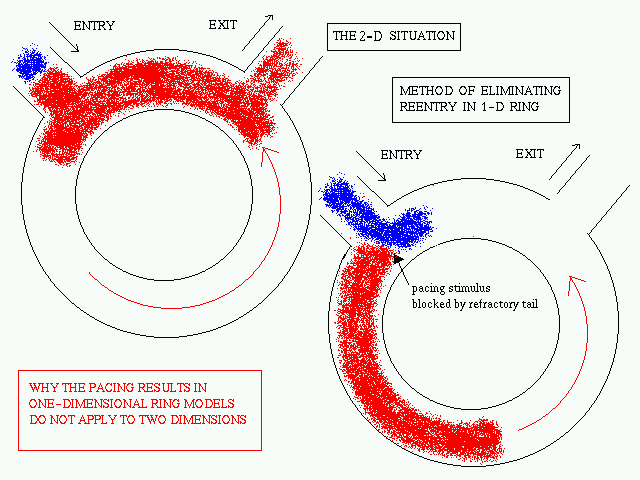
The pacing termination of reentry in a 1-D ring
is a well-studied problem. The termination of reentry occurs when the anterogarde
branch of the reentrant wave is blocked in a region which is still refractory
after the passage of the reentrant wave. Proper timing of the pacing wave
is essential as the anterograde branch must interact with the refractory
tail of the reentrant wave for successful termination to occur.
When the pacing site is not located on the 1-D
ring itself (as is generally the case in reality as well as the 2-D model)
this method of pacing termination of reentry fails in the absence of any
inhomogeneity. The reason for failure is explained below.
Revisiting an old problem : Reentry
in a One-dimensional Ring
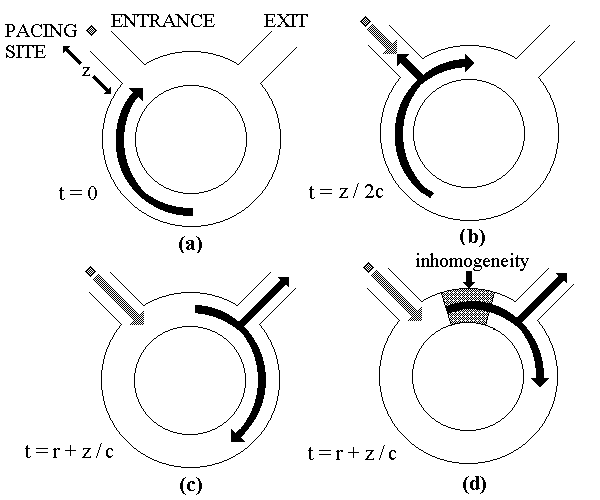
FIG. Schematic explanation of necessity
of inhomogeneity for successful pacing termination of reentry.
We consider a reentrant circuit as a 1D ring of
length L with separate entrance and exit sidebranches [a]. This
arrangement is an abstraction of the spatial geometry involved in anatomical
reentry. The pacing site is located on the entrance sidebranch at a distance
z from the circuit. We use the entrance sidebranch as the point
of spatial origin (x = 0) to define the location of the wave on
the ring. The conduction velocity and refractory period of the homogeneous
ring is assumed to be c and r, respectively. A pacing stimulus
is applied at time t = 0. This stimulus will collide with the branch of
the reentrant wave propagating out through the entrance sidebranch at t
= z/2c [b]. The pacing site will recover at t = r and if another stimulus
is applied immediately it will reach the reentry circuit at t = r + (z/c0
[c]. By this time the refractory tail of the reentrant wave will be at
a distance x = z away from the entrance sidebranch and the anterograde
branch of the stimulus will not be blocked. Thus,
when z > 0, it is impossible for the stimulus to catch up to the refractory
tail in a homogeneous medium. The situation
changes if an inhomogeneity (e.g., a zone of slow conduction) exists in
the circuit [d]. In this case, the above argument no longer holds because
the waves travel
faster or slower depending on their location
in the circuit. As a result, stimuli may arrive at the circuit from the
pacing site and encounter a region that is still refractory. This leads
to successful block of the anterograde branch of the stimulus, while the
retrograde branch annihilates the reentrant wave, resulting in successful
termination.
Effect of Inhomogeneity
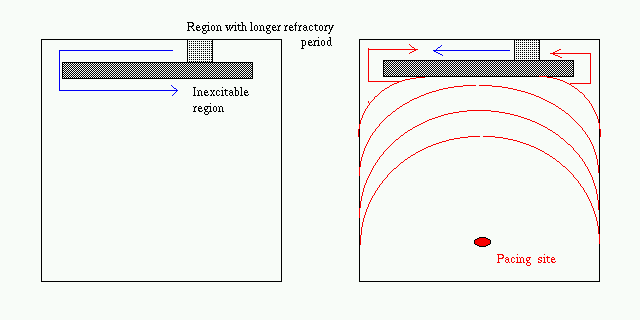
FIG. Assuming inhomogeneity - e.g., longer
refractory period or slower conduction in the narrow channel between non-conducting
obstacles may lead to successful blocking of the orthodromic (antegrade)
branch of the pacing wave. (Abildskov and Lux, 1995)
Effect of Anisotropy
Cardiac tissue shows anisotropic propagation
- the action potential propagates faster along the direction of the myocardial
fibers than transverse to it. The axis of anisotropy rotates along the
thickness of the myocardium (from the endocardial to the epicardial layer).
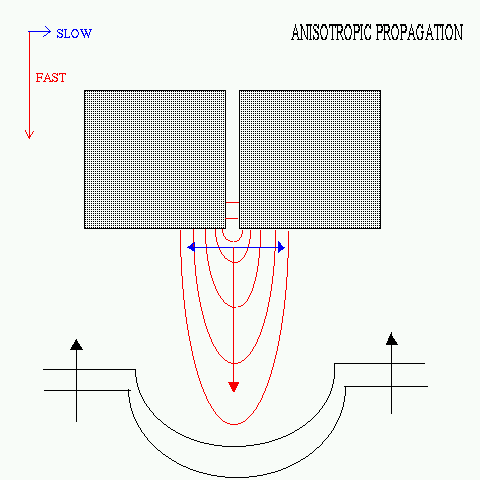 |
FIG. Anisotropic propagation. In
the present model the vertical axis is taken to be the fast axis as compared
to the horizontal axis with the diffusion coefficients along the respective
axes having a ratio of 1:0.3. (For comparison with actual physiological
values, note that the human ventricular myocardium shows longitudinal conduction
velocity of ~ 50 cm/sec and transverse conduction velocity of ~ 14 cm/sec.
[B. Surawicz, Electrophysiological Basis of ECG and Cardiac Arrhythmia,
p. 76])
Our simulations of pacing in such anisotropic models showed no qualitative difference from the results in isotropic models. Note that in multiple elctrode activation study if figure-of-eight reentry, it has been found that the narrow common return pathway is oriented along the fiber axis (i.e. longitudinal) [M. D. Lesh, J. F. Spear and E. N. Moore, in Zipes & Jalife, Cardiac Electrophysiology (1990), p. 374] |
Simulation Study
To understand the process of inhomogeneity-mediated
termination a 1D ring model is implemented.
We add a zone of slow conduction with conductance
differing from that of the remaining portion of the ring.
Length of zone, gradient at the boundary and
conductance value are varied to examine their effect on the propagation
of the anterograde branch of the extrastimuli.
Reentrant wave initiated at T = 0 by transient
conduction block over a part of the ring. Let the reentrant wave reach
at time T0 the site where the pacing wave will enter the circuit.
Two extrastimuli applied from the same point as the origin of the reentrant
wave :first extrastimulus at T1, second extrastimulus at T2
(coupling interval: T1-T0, pacing cycle: T2
- T1).
The first extrastimulus is able to terminate
reentry by itself if applied at the refractory tail of the reentrant wave
(the ``old'' 1D ring reentry problem). We avoid this as such a situation
is unlikely to occur if the pacing site is located away from the
reentry circuit.
Models used for simulation study:
2 variable models with Fitzhugh-Nagumo equations to describe the time-evolution
of the membrane potential (e) and a slow effective ionic conductance (g).
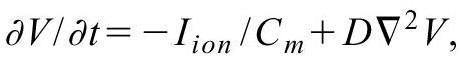
Compared to the effectiveness of rapid pacing
in eliminating anatomical reentry in actual cardiac tissue - in the modeling
studies pacing is effective only under assumption of the existence of a
region of slow conduction or a region with longer refractory period.
Results of the simulation study can be
seen from the figures available in the Simulation Results page.
Limitations of the present models:
1. 1- and 2-Dimensional - instead of 3-Dimensional
(the anisotropy axis rotates along the thickness)
2. Another method of simulating ischemia - increasing
K+ concentration (But this does not provide a chronic
arrhythmogenic substrate)
3. Monodomain assumed (this is justified for
the low-amplitude stimulus used in pacing)
The existence of inhomogeneities appears to be crucial for successful off-circuit pacing termination of anatomical reentry
Publications
Related Links